A major shift in sleep medicine is drawing attention across the United States as the first Sleep Apnea Drug approved for treatment begins changing how doctors approach one of the country’s most common sleep disorders. For decades, patients diagnosed with obstructive sleep apnea had limited options beyond machines, oral devices, or surgery. Now, new pharmaceutical therapies are entering the conversation and reshaping how millions may manage the condition.
The change began after U.S. regulators approved a medication specifically indicated for obstructive sleep apnea in adults with obesity. The development marked the first time a prescription drug was cleared to directly address the condition, opening the door for a completely new category of treatment.
Interest continues to grow as researchers test additional medications that could soon expand options even further.
The First FDA-Approved Sleep Apnea Drug
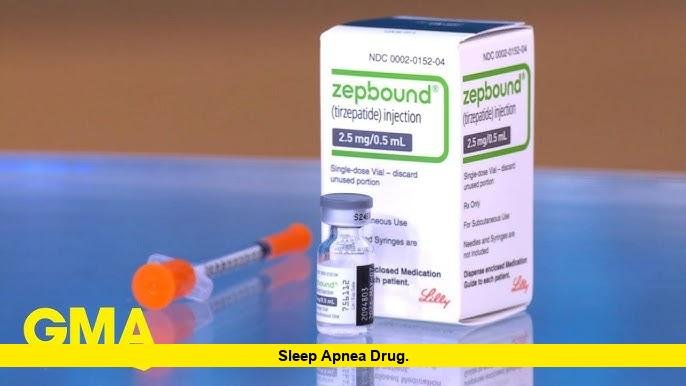
In December 2024, the U.S. Food and Drug Administration approved the medication tirzepatide—sold under the brand name Zepbound—for adults with moderate to severe obstructive sleep apnea who also have obesity.
This approval represented a historic milestone in sleep medicine. Until then, obstructive sleep apnea had never been treated with a prescription drug specifically approved for the disorder.
Tirzepatide was originally developed to treat type 2 diabetes and later gained widespread use as a weight-loss therapy. Researchers discovered that the drug’s metabolic effects could also help improve sleep apnea symptoms in patients whose condition is linked to excess body weight.
In clinical trials that supported approval, adults taking the medication experienced significant improvements in sleep apnea severity. Many participants had fewer breathing interruptions during sleep, and some saw their symptoms improve dramatically over time.
Doctors emphasize that the medication is intended to be used alongside lifestyle changes, including a reduced-calorie diet and increased physical activity.
How Tirzepatide Works for Obstructive Sleep Apnea
Obstructive sleep apnea occurs when throat muscles relax during sleep and block the airway. The repeated interruptions in breathing can lead to loud snoring, poor sleep quality, and severe daytime fatigue.
Excess weight is one of the most significant risk factors. Fat deposits around the neck can narrow the airway, making breathing interruptions more likely.
Tirzepatide addresses the underlying metabolic causes of obesity by targeting hormonal pathways involved in appetite and blood sugar regulation. By promoting significant weight loss, the medication can reduce pressure on the airway and decrease the number of breathing disruptions during sleep.
Studies showed that adults taking the medication often experienced fewer apnea episodes each hour during sleep. In some cases, symptoms were reduced enough that patients moved into a less severe category of the disorder.
Researchers say this indirect approach—treating the root cause rather than simply the airway blockage—represents a major evolution in sleep apnea treatment.
CPAP Machines Still Remain the Standard Treatment
Despite the excitement around medication options, sleep specialists stress that continuous positive airway pressure therapy remains the most effective treatment for many patients.
The CPAP machine uses a mask connected to a device that pushes air into the airway during sleep, preventing collapse and keeping breathing steady.
This method has been widely used since the early 1980s and is still considered the gold standard for treating obstructive sleep apnea when patients are able to tolerate it consistently.
However, adherence remains a challenge. Many patients struggle with discomfort, mask leakage, or difficulty sleeping with the machine.
Because of these challenges, researchers have been searching for alternative therapies that are easier for patients to maintain long term.
New Sleep Apnea Pill in Development
While tirzepatide is already approved, scientists are also studying a potential oral medication that could treat sleep apnea in a completely different way.
The experimental drug AD109 is designed as a once-daily pill that targets the neurological pathways controlling upper airway muscles. Instead of focusing on weight loss, the medication aims to strengthen airway muscle activity during sleep so the airway remains open.
The drug combines two compounds—atomoxetine and aroxybutynin—that increase signaling to airway muscles responsible for keeping the throat open.
Large clinical trials conducted across the United States have produced promising results.
In one phase 3 study, participants taking the medication experienced an average reduction of nearly 47% in their apnea-hypopnea index, a key measure used to assess sleep apnea severity.
Researchers also reported improvements in oxygen levels during sleep and significant reductions in nighttime breathing disruptions.
Because of these results, scientists are exploring the possibility that the pill could eventually become the first oral therapy specifically targeting the neuromuscular causes of sleep apnea.
Why Sleep Apnea Treatments Are Rapidly Evolving
Sleep apnea is far more common than many people realize. Health experts estimate that tens of millions of Americans experience the disorder, and many remain undiagnosed.
Untreated sleep apnea is linked to several serious health risks, including:
- High blood pressure
- Heart disease
- Stroke
- Type 2 diabetes
- Daytime fatigue and accidents
The condition occurs when breathing repeatedly stops during sleep, sometimes hundreds of times per night.
Because the disorder affects both sleep quality and cardiovascular health, researchers have spent years searching for treatments that address the root causes rather than simply managing symptoms.
The emergence of pharmaceutical therapies signals a broader shift in how the medical community approaches sleep disorders.
How Doctors Decide Who Can Use Medication
Not every patient diagnosed with sleep apnea will be prescribed medication.
The FDA approval for tirzepatide specifically applies to adults with moderate to severe obstructive sleep apnea who also have obesity.
Before prescribing the drug, physicians typically confirm the diagnosis with a sleep study and review the patient’s medical history.
Doctors may also recommend additional treatments alongside medication, such as:
- Weight management programs
- CPAP therapy
- Oral airway devices
- Lifestyle changes that improve sleep quality
Sleep specialists emphasize that treatment decisions depend on each patient’s individual condition and risk factors.
What This Means for the Future of Sleep Apnea Care
The introduction of pharmaceutical therapies could mark the beginning of a new era in sleep medicine.
Researchers believe that combining medication with lifestyle changes and traditional therapies may provide more personalized treatment plans for patients.
If ongoing clinical trials continue to show strong results, additional medications could reach the market in the coming years.
Some experts say these innovations could dramatically expand treatment options for people who struggle with existing therapies.
For millions of Americans who have spent years managing the disorder with nighttime machines, the arrival of new treatments is already changing the conversation around sleep apnea care.
Frequently Asked Questions About Sleep Apnea Drug Treatments
What is the first FDA-approved sleep apnea drug?
The first FDA-approved medication for obstructive sleep apnea is tirzepatide, marketed as Zepbound. It was approved in December 2024 for adults with moderate to severe obstructive sleep apnea who also have obesity.
How does a sleep apnea drug work?
Tirzepatide helps treat sleep apnea primarily by promoting weight loss. Reduced body weight can decrease pressure around the airway, which lowers the number of breathing interruptions during sleep.
Can medication replace CPAP therapy?
For many patients, CPAP therapy remains the most effective treatment. Medication may be used alongside other therapies depending on the patient’s medical condition.
Are there new sleep apnea pills being developed?
Yes. A medication known as AD109 is currently undergoing clinical trials and has shown promising results in reducing sleep apnea severity in study participants.
How common is obstructive sleep apnea?
Millions of Americans live with obstructive sleep apnea. Many cases remain undiagnosed because symptoms often occur during sleep.
What do you think about the rise of medication-based treatments for sleep apnea? Share your thoughts or follow the latest updates as new therapies continue to emerge.